Last Updated on 06/09/2022 by qhbake
Se sei interessato a vendere le tue caramelle gommose probiotiche, probabilmente sei curioso del processo. In questo articolo discuteremo gli ingredienti del prodotto, il processo di produzione e il programma di controllo della qualità. Parleremo anche della data di scadenza. Alla fine di questo articolo, dovresti avere un'idea migliore di come scegliere la migliore marca di caramelle gommose probiotiche.
ingredienti
La qualità delle caramelle gommose probiotiche inizia dal fornitore del ceppo e si estende fino alla confezione finale. Un imballaggio scadente compromette la formulazione, influenzandone la potenza e le prestazioni. I produttori devono affrontare queste sfide. Un imballaggio inefficace richiede un'eccedenza significativa per compensare la perdita di principi attivi. Questo, a sua volta, aumenta il costo di produzione per dose. Di seguito sono elencati gli ingredienti che compongono le migliori caramelle gommose probiotiche prodotte in fabbrica.
Quando si producono caramelle gommose probiotiche, è importante seguire rigorosi standard di controllo della qualità. I batteri probiotici devono essere stabili e soddisfare la dichiarazione di etichetta per lotto. Inoltre, l'ambiente di fabbrica deve essere pulito e privo di allergeni e impurità. Il produttore dovrebbe evitare di utilizzare terreni di coltura o altri additivi che potrebbero danneggiare i ceppi probiotici. Per garantire qualità e coerenza, un produttore dovrebbe scegliere una struttura che mantenga rigorose procedure di controllo della qualità.
Processo di produzione
Un produttore deve scegliere con cura ceppi naturali di batteri resistenti agli acidi e alla bile nel corpo umano. Dopo aver identificato questi ceppi, il produttore deve determinare come mantenere la loro stabilità e potenza. A seconda del ceppo, vengono utilizzati diversi metodi di essiccazione per proteggerne la vitalità. La liofilizzazione è un processo più delicato mentre l'essiccazione a spruzzo è un metodo più veloce. Il processo di produzione delle caramelle gommose probiotiche è diverso dalla produzione di altri integratori alimentari.
Il produttore deve considerare il tipo di consegna del prodotto. Le caramelle gommose contengono elevate quantità di umidità, che non è un ambiente favorevole per i probiotici tradizionali. Tuttavia, le caramelle gommose sono popolari tra gli adulti che amano i dolci. Oltre al contenuto di zucchero, la maggior parte dei prodotti gommosi elenca lo zucchero come ingrediente. Sebbene lo zucchero non abbia valore nutrizionale, contribuisce all'obesità, alla carie e al diabete. Alcuni prodotti hanno anche tappi a prova di bambino per evitare il consumo accidentale.
Programma di controllo della qualità
La creazione di un programma di controllo della qualità per le caramelle gommose probiotiche che possono essere vendute come integratori a marchio privato inizia con il test delle materie prime per i batteri. L'approccio più basilare consiste nell'iniziare con più batteri del necessario, supponendo che alcuni di questi periscano prima che il prodotto raggiunga le mani del consumatore. Ma i test di stabilità possono aiutarti a determinare la giusta quantità di batteri ed è fondamentale includere questo passaggio nel processo di produzione.
Oltre a testare le materie prime, un programma di controllo qualità per le caramelle gommose probiotiche prodotte da una fabbrica dovrebbe includere anche la valutazione delle capacità del processo di produzione. Il processo di produzione può essere automatizzato per eliminare l'errore umano, ma è comunque importante tenere d'occhio la qualità. Per fare questo, la formazione degli operatori è fondamentale. Anche il turnover dei dipendenti deve essere basso. Inoltre, gli approcci Six Sigma possono essere utilizzati per misurare la qualità del prodotto e migliorare continuamente il processo.
Data di scadenza
Se stai assumendo un integratore probiotico, controlla la data di scadenza. I probiotici sono microrganismi vivi che possono essere utili per una varietà di condizioni. In buone condizioni di conservazione, possono rimanere efficaci da 18 a 24 mesi. Tuttavia, se non apri subito i tuoi probiotici, perderanno la loro potenza. Il modo migliore per evitare integratori probiotici scaduti è acquistarne di freschi.
Un produttore rispettabile rispetterà la data di scadenza dell'etichetta, nonché la salute e la sicurezza dei suoi ingredienti. Il produttore dovrebbe disporre di un processo collaudato per incorporare i probiotici nella matrice della gomma e nel rivestimento zuccherino. Ciò garantisce un elevato recupero dopo la produzione e una solida stabilità per almeno 18 mesi. Inoltre, il produttore dovrebbe essere in grado di fornire una convalida indipendente da parte di terzi, che assicurerà che i suoi prodotti siano privi di contaminazione.
-

Linea di deposito automatico di caramelle gommose senza amido da 150: 60.000 caramelle gommose/ora, 280 stampi all'interno della macchina, unità di raffreddamento 8-10PH
-
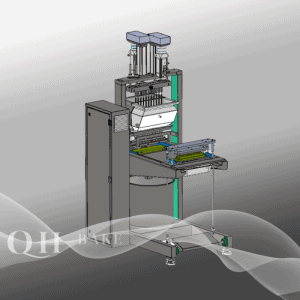
Attrezzatura per il deposito di caramelle Servo-Form 20S: Cavità PCS: ±20; 100x80x160 Dimensioni (cm)
-

Linea di deposito automatico di caramelle gommose senza amido da 300.000 caramelle gommose/ora, 8-10 PH, unità di raffreddamento da 280 pezzi con doppio stampo all'interno della macchina.
-

50S Servo Control Candy Depositor: Servo; Cavità PCS: ±20; Dimensioni 200x100x170 (cm)
-

Linea di deposito automatico di caramelle gommose senza amido 600: 240.000 caramelle gommose/ora 10-20 PH Unità di raffreddamento 520 pezzi di doppio stampo all'interno della macchina
-

80 Linea di produzione automatica di caramelle gommose: 40.000 caramelle gommose/h Unità di raffreddamento 5-8PH 200 pezzi di stampo all'interno della macchina
-

Macchina automatica per la produzione di perle Konjac per la vendita - Fornitore e produzione
-

Linea di produzione automatica di lecca-lecca in vendita - Fabbrica di macchine per dolci
-

Linea di produzione automatica con deposito senza amido: rivoluziona la produzione di caramelle